Dr. Peter J. Skabara, Professor at the Department of Pure and Applied Chemistry at the University of Strathclyde (Glasgow, UK) presents a lecture entitled, "The Role of Non-covalent Interactions in Band-gap Tuning, Conformation and Self-assembly" as part of the Georgia Tech-COPE Seminar Series.
Abstract
The extent to which the optoelectronic properties of molecules are controlled by their conformation and packing is now well appreciated. In particular, the planarity (or nonplanarity) of a molecule has a dramatic influence on its behavior, and hence on its potential application. In the context of light emission, disorder can be beneficial in controlling aggregation, but for photovoltaics and transistors where low energy absorption and effective charge transport are key issues, planar molecules are preferable. Of particular relevance to this talk is the development of planarised conjugated architectures that should support much-enhanced charge transport properties as a result of their ability to self-assemble into tightly packed frameworks.
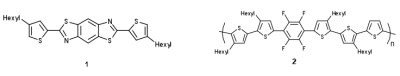
For example, compound 1 (m = 10-2 cm2 V-1 s-1) orders into a 3-dimensional network of close contacts both in crystalline form and as a thin film. This high level of self-assembly is achieved through p-p stacking in 2D and non-covalent interactions between the S and N atoms of adjacent molecules in the third. The nature of these interactions is not entirely clear, but one possible source of an attractive potential is a 3c-2e interaction between lone pairs and relatively low-lying antibonding orbitals, although these are likely to be counteracted to some extent by significant lone pair-lone pair repulsions. The 3D nature of the material represents a unique motif for highly efficient charge transport. A second example, polymer 2 (m = 10-3 cm2 V-1 s-1), has a planar repeat unit in the solid state as a result of close intramolecular sulfur/fluorine and hydrogen/fluorine contacts. Interestingly, variations in the absorption spectrum of these molecules suggest that the band gap is very sensitive to the degree of planarity. Whilst the mobility values for 1 and 2 are modest, the importance of the work is that it establishes a principle for the rational design of second generation materials with enhanced charge-transport properties.
This lecture will discuss several further examples that underline the importance and influence of non-covalent interaction in organic semiconductors.